EIGHTEENTH ISSUE
September 7, 2022
sk(in depth) scoop
And, another one? Just this week, yet another biologic was FDA approved for the treatment of psoriasis. Spesolimab-sbzo, better known as SPEVIGO, is now the first U.S. FDA-approved therapy for generalized pustular psoriasis (GPP) flares in adults. GPP is a distinct entity from other types of psoriasis, including plaque psoriasis, and can result in severe, life-threatening disease. SPEVIGO, a selective antibody which blocks the activity of IL-36, a key signaler in the GPP inflammatory pathway, represents a revolutionary step towards improved care of this subset of psoriatic patients.
So what’s the big deal?
What did the clinical trial show?
Main takeaway: The U.S. FDA’s approval of SPEVIGO for generalized pustular psoriasis represents another important step in the development of targeted therapies for severe dermatologic disease.
So what’s the big deal?
- GPP can result in life threatening complications; however, because of its rarity, recognizing its signs and symptoms may lead to delays in diagnosis
- Though there are a variety of drugs that have been developed to treat plaque psoriasis and psoriatic arthritis, these drugs mostly target IL-17 and IL-23, and are not specifically indicated for control of GPP flares
What did the clinical trial show?
- Among 53 patients experiencing a GPP flare, 54% treated with SPEVIGO showed no visible pustules at 1 week, compared to 6% of those administered placebo
- Side effects included drug reaction in 2 patients and infection in 17% through the first week
Main takeaway: The U.S. FDA’s approval of SPEVIGO for generalized pustular psoriasis represents another important step in the development of targeted therapies for severe dermatologic disease.
Just keep climbing!
Metastatic melanoma used to be a worst case scenario diagnosis. However, the development of immune checkpoint inhibitors and BRAF/MEK inhibitors has changed that, transforming management of metastatic melanoma. Immune checkpoint inhibitors (ICIs) refer to therapies that block “checkpoints” in the immune system, thereby stimulating the body’s T-cells to attack cancer cells. Common ICIs include anti–programmed cell death 1 (anti–PD-1) antibodies, nivolumab and pembrolizumab, and the anti–CTLA-4 antibody, ipilimumab. With the introduction of ICIs, along with BRAF and MEK inhibitors, the overall survival (OS) rate for metastatic melanoma rose from 10-30% to 50%. In 2015 the National Cancer Comprehensive Network (NCCN) recommended anti-PD-1 ICIs as first-line therapy for patients with metastatic melanoma; it was subsequently FDA approved. Since then, anti-PD-1 ICI monotherapy of nivolumab and pembrolizumab or combined anti-PD-1 treatments with anti-CTLA-4 therapy (ipilimumab) have been recommended. However, it is unclear how often these first-line treatments are actually used for metastatic melanoma.
This study aimed to discover the overall utilization rates and outcomes of ICI as a first line treatment for metastatic melanoma. Using the National Cancer Database, this retrospective cohort study included 16,831 adult patients with newly diagnosed Stage IV cutaneous melanoma between 2010 and 2019.
Key findings:
Limitations: This study was limited in that it was retrospective in nature. Additionally the data studied was >3 years old and the use of ICIs may have increased since 2019.
Main Takeaway: While the use of immune checkpoint inhibitors for metastatic melanoma have increased in response to guidelines, it is underutilized in patients with lower socio-economic status and those with brain metastases. Ongoing efforts to utilize ICIs as first-line treatment for metastatic melanoma are warranted.
Metastatic melanoma used to be a worst case scenario diagnosis. However, the development of immune checkpoint inhibitors and BRAF/MEK inhibitors has changed that, transforming management of metastatic melanoma. Immune checkpoint inhibitors (ICIs) refer to therapies that block “checkpoints” in the immune system, thereby stimulating the body’s T-cells to attack cancer cells. Common ICIs include anti–programmed cell death 1 (anti–PD-1) antibodies, nivolumab and pembrolizumab, and the anti–CTLA-4 antibody, ipilimumab. With the introduction of ICIs, along with BRAF and MEK inhibitors, the overall survival (OS) rate for metastatic melanoma rose from 10-30% to 50%. In 2015 the National Cancer Comprehensive Network (NCCN) recommended anti-PD-1 ICIs as first-line therapy for patients with metastatic melanoma; it was subsequently FDA approved. Since then, anti-PD-1 ICI monotherapy of nivolumab and pembrolizumab or combined anti-PD-1 treatments with anti-CTLA-4 therapy (ipilimumab) have been recommended. However, it is unclear how often these first-line treatments are actually used for metastatic melanoma.
This study aimed to discover the overall utilization rates and outcomes of ICI as a first line treatment for metastatic melanoma. Using the National Cancer Database, this retrospective cohort study included 16,831 adult patients with newly diagnosed Stage IV cutaneous melanoma between 2010 and 2019.
Key findings:
- The use of first-line ICIs increased from 8.9% in 2010 to 38.8% in 2015 and 62.5% in 2019
- Overall survival was longer for those using ICIs: 43.7 months in ICI group vs. 16.1 months for targeted therapy or chemotherapy group
- Similar significant improvement in overall survival was seen in ICI treated patients with melanoma metastases to the brain (19.9 months) compared with other treatments (10.7 months). The difference was not significant in melanoma patients with metastasis to the liver
- Subpopulation analysis showed that older patients, those with Medicaid insurance, and people from zip codes with lower median household incomes, and who had brain metastases were less likely to receive ICI as a first line treatment
Limitations: This study was limited in that it was retrospective in nature. Additionally the data studied was >3 years old and the use of ICIs may have increased since 2019.
Main Takeaway: While the use of immune checkpoint inhibitors for metastatic melanoma have increased in response to guidelines, it is underutilized in patients with lower socio-economic status and those with brain metastases. Ongoing efforts to utilize ICIs as first-line treatment for metastatic melanoma are warranted.
Imiquimod and 5-FU treatment are superior to ALA with PDT in the prevention of cSCC 1 year post-treatment
Journal of the American Academy of Dermatology
Journal of the American Academy of Dermatology
It matters how you treat your AKs!
Actinic keratoses (AKs) are precancerous lesions that can transform into cutaneous squamous cell carcinoma (cSCC). Some treatment options for extensive AKs include 5-fluorouracil (5-FU), imiquimod, and photodynamic therapy with aminolevulinic acid (PDT-ALA). Researchers sought to compare the long-term efficacy of 5-FU, imiquimod, and PDT-ALA. They used the amount of time post-treatment until the development from AK to cSCC as the primary outcome.
In this retrospective cohort study, 78,878 patients ≥ 18 years old were identified in the Optum Clinformatics Data Mart database. Patients diagnosed with AKs who were subsequently prescribed 5-FU or imiquimod or who underwent PDT-ALA treatment were identified. Results showed the rate of cSCC development was not significantly different between patients treated with 5-FU and patients treated with imiquimod (HR 0.99; 95% CI, 0.90- 1.08). However, the study found that patients treated with PDT-ALA had a significantly higher rate of cSCC development compared to imiquimod and 5-FU patients (HR 1.29; 95% CI, 1.17-1.43).
Limitations: Equating AK destruction with cSCC prevention may not be appropriate. The location of field therapy could not be determined.
Main Takeaway: While 5-FU did NOT demonstrate superiority to imiquimod, Imiquimod and 5-FU demonstrated superiority to PDT-ALA in the prevention of AK transformation to cSCC >1 year post-treatment.
Actinic keratoses (AKs) are precancerous lesions that can transform into cutaneous squamous cell carcinoma (cSCC). Some treatment options for extensive AKs include 5-fluorouracil (5-FU), imiquimod, and photodynamic therapy with aminolevulinic acid (PDT-ALA). Researchers sought to compare the long-term efficacy of 5-FU, imiquimod, and PDT-ALA. They used the amount of time post-treatment until the development from AK to cSCC as the primary outcome.
In this retrospective cohort study, 78,878 patients ≥ 18 years old were identified in the Optum Clinformatics Data Mart database. Patients diagnosed with AKs who were subsequently prescribed 5-FU or imiquimod or who underwent PDT-ALA treatment were identified. Results showed the rate of cSCC development was not significantly different between patients treated with 5-FU and patients treated with imiquimod (HR 0.99; 95% CI, 0.90- 1.08). However, the study found that patients treated with PDT-ALA had a significantly higher rate of cSCC development compared to imiquimod and 5-FU patients (HR 1.29; 95% CI, 1.17-1.43).
Limitations: Equating AK destruction with cSCC prevention may not be appropriate. The location of field therapy could not be determined.
Main Takeaway: While 5-FU did NOT demonstrate superiority to imiquimod, Imiquimod and 5-FU demonstrated superiority to PDT-ALA in the prevention of AK transformation to cSCC >1 year post-treatment.
Patients receiving adalimumab combined with methotrexate for psoriasis resulted in decreased drug clearance and fewer antidrug antibodies than adalimumab alone
Journal of Investigative Dermatology
Patients receiving adalimumab combined with methotrexate for psoriasis resulted in decreased drug clearance and fewer antidrug antibodies than adalimumab alone
Journal of Investigative Dermatology
We all get by with a little help from our friends… and methotrexate!
Adalimumab is a TNF-alpha inhibitor used in treatment of psoriasis, but its use can be limited by development of 1) antidrug antibodies that inhibit the drug binding to TNF-alpha and 2) development of immune complexes, which promote clearance of the drug. In rheumatoid arthritis, adding methotrexate (adalimumab + methotrexate) can limit antidrug antibody formation and decrease drug clearance, thus improving the clinical response. Small observational studies suggest promising clinical results in treating psoriasis with both adalimumab and methotrexate. However, the long-term effects of the combination of these two medications has not been studied in psoriasis.
In this study, the authors conducted a single-blind randomized controlled trial to examine the efficacy and safety of two drugs, adalimumab + methotrexate, versus adalimumab monotherapy in treatment of chronic plaque psoriasis. Thirty-one patients with plaque psoriasis received adalimumab + methotrexate, and 30 received adalimumab alone. Overall, no serious adverse events were reported. The authors found that after 1 year, the group receiving adalimumab + methotrexate had improved PASI scores (psoriasis area and severity index, a tool to measure severity and extent of psoriasis), with the mean score in the group receiving both drugs being 58.1 versus 36.7 in the monotherapy group. Additionally, there was better drug survival (i.e. less drug clearance) in the group receiving both medications, with serum-trough concentrations at 6.8 versus 5.9 in the monotherapy group. Furthermore, fewer patients had antidrug antibodies in the group who received both adalimumab and methotrexate (22.6%) compared to the group receiving adalimumab alone (60%).
Limitations of this study include a relatively small sample size and the lack of longer-term followup data (>1 year).
Main Takeaway: Patients with plaque psoriasis receiving both adalimumab and methotrexate developed less antidrug antibodies, had improved PASI scores, and demonstrated prolonged drug survival.
Efficacy and safety of topical brepocitinib for the treatment of mild-to-moderate atopic dermatitis
British Journal of Dermatology
Just another Janus kinase inhibitor? Not so fast!
We all know that janus kinase (JAK) inhibitors are the hot new kids on the block for treating atopic dermatitis, a common inflammatory skin disease characterized by pruritus. The JAK family of kinases, including JAK1, JAK2, JAK3, and tyrosine kinase 2 (TYK2), are often implicated in activation of the inflammatory cascade. Recently approved JAK inhibitors target JAK1 or JAK1 and 2, however brepocitinib is the first to inhibit JAK1 and TYK2.
This phase IIb, double-blind study randomized 292 patients to receive one of eight treatments for 6 weeks:
- Brepocitinib 0.1% once daily
- Brepocitinib 0.3% once daily
- Brepocitinib 0.3% twice daily
- Brepocitinib 1% once daily
- Brepocitinib 1% twice daily
- Brepocitinib 3% once daily
- Placebo once daily
- Placebo twice daily
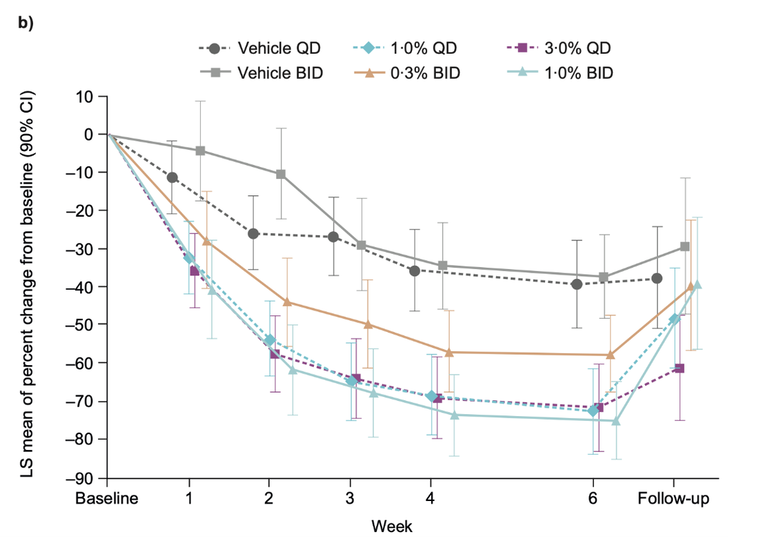
Percent change in Eczema Area and Severity Score (EASI) from baseline to week 6, as well as adverse events, were monitored. Reduction in EASI score at week 6 was significantly greater in the groups receiving brepocitinib 1% dosed once daily (-70.1) or twice daily (-75.0) (p-value <0.05). This reduction was evident as early as week 1 (Figure 1). No serious adverse events or deaths occurred.
Limitations: As this was a dose-ranging study, sample sizes of the individual treatment groups were small. Effect sizes should be confirmed in larger studies with longer treatment times.
Main take-away: Topical brepocitinib 1% is a safe and effective treatment for symptoms in patients with mild-to-moderate atopic dermatitis.
Main take-away: Topical brepocitinib 1% is a safe and effective treatment for symptoms in patients with mild-to-moderate atopic dermatitis.
Clinical Outcomes of Small Infantile Hemangiomas Treated With Pulsed Dye Laser
Dermatologic Surgery
To laser or to let be.
Infantile hemangiomas are benign vascular tumors that affect up to 5% of infants. This study looked at the residual skin effects of pulsed dye laser (PDL) treatment on 58 small, low-risk infantile hemangiomas (i.e. high-risk hemangiomas can include locations on the eyes, nose and neck, associations with possible airway compromise, and any >2 cm).
The study found that PDL treatment led to statistically significant increases in anetoderma (characterized by wrinkled outpouchings in the skin) and fibrofatty tissue (p=.036 and .033). Prior studies have also demonstrated that 54% of patients undergoing PDL have subsequently experienced cosmetic problems such as hypopigmentation and cutaneous atrophy.
Limitations: This research investigated a small cohort without a control group of patients with infantile hemangiomas who went with the “let it be” approach and did not undergo treatment.
Main Takeaway: Pulsed dye laser, a common treatment for low-risk infantile hemangiomas, led to residual skin changes such as anetoderma and fibrofatty tissue.
Infantile hemangiomas are benign vascular tumors that affect up to 5% of infants. This study looked at the residual skin effects of pulsed dye laser (PDL) treatment on 58 small, low-risk infantile hemangiomas (i.e. high-risk hemangiomas can include locations on the eyes, nose and neck, associations with possible airway compromise, and any >2 cm).
The study found that PDL treatment led to statistically significant increases in anetoderma (characterized by wrinkled outpouchings in the skin) and fibrofatty tissue (p=.036 and .033). Prior studies have also demonstrated that 54% of patients undergoing PDL have subsequently experienced cosmetic problems such as hypopigmentation and cutaneous atrophy.
Limitations: This research investigated a small cohort without a control group of patients with infantile hemangiomas who went with the “let it be” approach and did not undergo treatment.
Main Takeaway: Pulsed dye laser, a common treatment for low-risk infantile hemangiomas, led to residual skin changes such as anetoderma and fibrofatty tissue.
Topical ectoine and hyaluronic acid cream for pediatric patients with atopic dermatitis
Pediatric Dermatology
Topical ectoine and hyaluronic acid cream for pediatric patients with atopic dermatitis
Pediatric Dermatology
Could ectoine and hyaluronic acid cream be the new ~hyal-oly grail~ for atopic dermatitis?!
Atopic dermatitis (AD), or eczema, is a chronic inflammatory disease characterized by intense pruritus that commonly affects infants and children. The disease often follows a chronic relapsing course with a significant impact on patients’ quality of life. Although numerous treatments are available, many are associated with adverse effects due to long-term usage.
Ectoine is a low-molecular-weight, water-binding, organic osmolyte. In previous studies, topical application of ectoine to healthy human skin improved skin hydration as well as skin barrier function. In addition, 1% ectoine and 0.1% hyaluronic acid (EHA) containing cream improved skin lipid barrier function and restored dry, sensitive atopic skin in adults. Here, the authors studied the safety and efficacy of EHA cream in a placebo-controlled, randomized study of 70 patients with AD between the ages of 2 and 18.
The authors assessed outcomes and safety by comparing changes in the SCORAD index (a clinical tool used to assess the extent and severity of eczema) between the EHA cream vs vehicle cream (placebo) group. By week 4, patients receiving EHA cream had significant clinical improvement based on SCORAD (mean difference in change from baseline score was -6.62) compared to patients in the placebo group. Patients receiving EHA cream also experienced a greater reduction of pruritus (mean difference in change from baseline to intermediate visit was -2.40). In terms of adverse events, 8 cutaneous-related adverse events including skin erythema, pruritus, and burning sensation were reported in EHA cream, compared to two adverse events in vehicle cream.
Given the relatively short treatment period, future studies encompassing a longer duration of treatment are needed to evaluate the long-term efficacy and safety of EHA cream.
Main Takeaway: This study found that ectoine 1% and hyaluronic acid 0.1% cream may be an effective and promising agent for pediatric patients with mild-to-moderate AD.
Atopic dermatitis (AD), or eczema, is a chronic inflammatory disease characterized by intense pruritus that commonly affects infants and children. The disease often follows a chronic relapsing course with a significant impact on patients’ quality of life. Although numerous treatments are available, many are associated with adverse effects due to long-term usage.
Ectoine is a low-molecular-weight, water-binding, organic osmolyte. In previous studies, topical application of ectoine to healthy human skin improved skin hydration as well as skin barrier function. In addition, 1% ectoine and 0.1% hyaluronic acid (EHA) containing cream improved skin lipid barrier function and restored dry, sensitive atopic skin in adults. Here, the authors studied the safety and efficacy of EHA cream in a placebo-controlled, randomized study of 70 patients with AD between the ages of 2 and 18.
The authors assessed outcomes and safety by comparing changes in the SCORAD index (a clinical tool used to assess the extent and severity of eczema) between the EHA cream vs vehicle cream (placebo) group. By week 4, patients receiving EHA cream had significant clinical improvement based on SCORAD (mean difference in change from baseline score was -6.62) compared to patients in the placebo group. Patients receiving EHA cream also experienced a greater reduction of pruritus (mean difference in change from baseline to intermediate visit was -2.40). In terms of adverse events, 8 cutaneous-related adverse events including skin erythema, pruritus, and burning sensation were reported in EHA cream, compared to two adverse events in vehicle cream.
Given the relatively short treatment period, future studies encompassing a longer duration of treatment are needed to evaluate the long-term efficacy and safety of EHA cream.
Main Takeaway: This study found that ectoine 1% and hyaluronic acid 0.1% cream may be an effective and promising agent for pediatric patients with mild-to-moderate AD.